Structural Approach to Ribosome Biogenesis
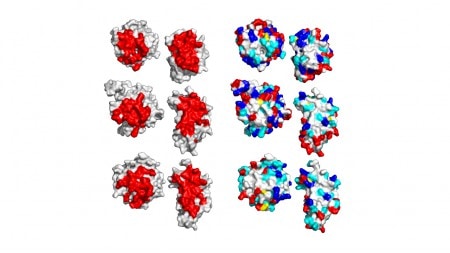
The extraordinary plasticity of Trm112 makes it a versatile methyl transferase co-activator interacting with modification enzymes involved in rRNA, tRNA and protein methylation.” In collaboration with Marc Graille (Ecole Polytechnique, Palaiseau)
In collaboration with structural biologists, we are characterizing the 3-D structure of assembly factors involved in ribosome biogenesis.
Many ribosome assembly factors have been identified over the years and the field is now facing the daunting task to assign them a precise function. This is complicated because most factors do not carry any obvious protein motifs in their sequence. In this context, structural approaches have proven extremely powerful to understand what assembly factors do. CryoEM analyses of precursor ribosome complexes have revealed that many ribosome assembly factors act to protect, and possibly proofread, functional ribosome sites during biogenesis. High-resolution structure determination of individual assembly factors, and subcomplexes of factors, has started to reveal their function.

